ABOUT ROOSTERBIO
RoosterBio accelerates human mesenchymal stem/stromal cell (hMSC) and extracellular vesicle (EV) product and process development to fuel the rapid commercialization of scalable regenerative cures. Our high-quality hMSCs, bioprocess media, genetic engineering tools, and EV production solutions are paired with expert bioprocessing knowledge to progress therapeutic developers from concept to first-in-human testing and commercial manufacturing at reduced cost and increased productivity. With optimized, scalable processes, Type 2 Drug Master Files, and cGMP products, we have enabled therapeutic programs to traverse their path to clinical translation in under 1 year.
RoosterBio is driven by our client's success and creating a world where safe and effective regenerative medicines are rapidly developed and widely available on a global scale.
FEATURED ARTICLES
-
MISEV guidelines help extracellular vesicle researchers design robust experiments through transparent reporting and rigorous characterization rather than rigid checklists.
-
Filter fouling has limited exosome therapeutic scalability—until now. Explore how a breakthrough reagent achieves 50% particle recovery versus 5% traditionally, enabling affordable clinical manufacturing.
-
See how two academic investigators accelerated corneal regeneration and lung fibrosis therapies from preclinical research to clinical trials by leveraging scalable GMP solutions and regulatory-ready materials.
-
Explore how bone marrow, umbilical cord, and adipose MSCs produce extracellular vesicles with distinct characteristics, and why 3D culture systems consistently outperform 2D platforms in productivity.
-
See how downstream processing affects CD73 expression and function on MSC-EVs through orthogonal analytical methods that connect vesicle content to biological activity and therapeutic potential.
-
Understand how a downstream treatment prevents filter fouling during exosome purification, enabling 40-70% particle recovery compared to traditional 5-10% yields while maintaining quality attributes.
-
Explore how chemically defined media and optimized bioreactor protocols increase exosome yields 40-fold while reducing commercial manufacturing costs from $10,000 to under $1,000 per dose.
-
What separates fibroblasts from MSCs when both express identical markers and differentiate into bone, fat, and cartilage? Molecular profiling reveals they exist on a functional spectrum of plasticity.
-
Explore comprehensive analytics comparing MSC-EV quality attributes across tissue sources, donors, and scalable production platforms to identify critical manufacturing variables.
-
Explore quantitative single-vesicle profiling methods that reveal tissue-specific surface marker expression patterns invisible to bulk EV analysis, with molecular-level resolution across three MSC sources.
-
Understand how single-vesicle analysis exposes critical EV heterogeneity, why MSC-EVs face manufacturing hurdles, and what regulators expect from developers pursuing clinical translation.
-
Explore how hMSCs power organ-on-chip systems through multipotency and scalability—from Georgia Tech's bone marrow models to ISS microgravity experiments and precision bioprinting innovations.
-
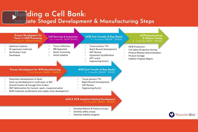
Understand how early cell banking strategy and manufacturing platform selection influence regulatory timelines, cost structures, and scalability from Phase I through commercial production for MSC therapies.
-
What separates genuine exosomes from microvesicles, apoptotic bodies, or lipoproteins? Explore the markers, assays, and biogenesis pathways that define these particles beyond popular terminology.
-
Uncover strategies to reduce cell bank development from years to months. See how GMP-compliant partnerships eliminate redundant documentation and accelerate your path to first-in-human trials.
-
Senescent cells spread inflammation and fibrosis across tissues. Learn how MSC-derived exosomes and secreted factors reverse these aging hallmarks by transferring fresh mitochondria and suppressing inflammatory cascades.
-
Understand why IV-delivered MSCs undergo rapid apoptosis in lung tissue while locally implanted cells survive as programmable therapeutic factories—and how this knowledge enables rational MSC engineering.
-
Discover how effective cell banking and scale-up strategies can reduce risk and cost in advanced therapy manufacturing. Learn practical ways to meet increasing cell and exosome quantity needs.
-
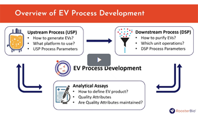
Uncover how analytical tools assess the impact of production process choices and tissue sources on the quality of cell-derived extracellular vesicles (EVs).
-
Biological components in conditioned media are a consistent problem during downstream processing of EVs. A novel in-process reagent prevents these issues and improves particle recovery 10-fold while maintaining CQAs.
CONTACT INFORMATION
RoosterBio
5295 Westview Drive Suite 275
Frederick, MD 21703
UNITED STATES
Phone: 240-831-4914
RESOURCES
-
When MSCs are genome-edited, it will inevitably be done with cell therapy in mind and should involve complementary breakthroughs to scale up and industrialize gene delivery.
-
RoosterVial™-hDF working cell banks contain cryopreserved xeno-free primary human dermal fibroblasts (hDFs) isolated from normal neonatal foreskin. These cell banks are manufactured with highly standardized processes and supported by first-in-class characterization for R&D and product development.
-
Available in multiple tissues origins and from multiple donors, RoosterBio’s human mesenchymal stem/stromal cells (hMSCs) are sourced under strict guidelines, manufactured with highly standardized processes, and supported by 1st in class characterization.
-
Eliminate media exchanges with RoosterNourish™ hMSC expansion medium and RoosterReplenish™ in-process bioreactor feed.
-
Standardize the production of exosomes/extracellular vesicles with a translation-friendly, scalable system designed to streamline your process from cell-expansion to optimized EV collection yields.
-
RoosterGEM™ is a complete genetic engineering medium that allows researchers and product developers to confidently engineer their therapeutic product with simplified end-to-end processes.
-
RoosterBio’s Cell & Media Kits allow you to economically screen multiple hMSC donors or tissues and rapidly evaluate RoosterBio’s media exchange-free system.
-
RoosterBio’s CliniControl™ products include human mesenchymal stem/stromal cells, bioprocess expansion media, and exosome/extracellular vesicle production media manufactured in compliance with cGMPs and supported by U.S. FDA Type II Master Files.
-
The path to IND approval for cell-based therapies can be challenging and time-consuming with strict regulatory requirements.
-
Advanced therapy programs are complex arrays of upstream and downstream processes that must reliably work together to produce consistent products worthy of clinical use.
BROCHURES
- Xeno-Free (XF) Human Dermal Fibroblasts
- RoosterCollect EV Pro™ - Extracellular Vesicle (EV) Production
- RoosterGEM™ - A Complete Genetic Engineering Medium
- RoosterReplenish™ hMSC Bioreactor Feed
- RoosterNourish™ MSC Expansion Media
- RoosterVial™ Xeno-Free MSCs
- Ready-To-Use Exosomes That Streamline R&D
- RoosterCollect™ Exosome/Extracellular Vesicle Production
- RoosterBio Process Development Services