
RECENT WEBINARS

How Connected Manufacturing Operations And AI Optimize Production
While 53% of manufacturers are digital, a lack of system connectivity stalls AI progress. Learn how to break down data silos to reduce rework, accelerate quality reviews, and ensure AI readiness.

Advancing In-Line PAT Solutions For Real-Time Batch Analysis
Shift from reactive sampling to continuous, data-driven control. Learn how in-line metabolic monitoring improves yield and batch consistency through real-time glucose and lactate insights.
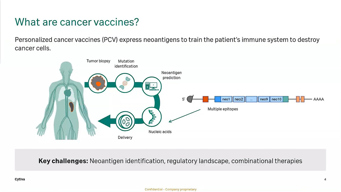
Advancing mRNA-LNP Delivery And Small-Scale Manufacturing, APAC Session
Learn how ionizable LNP formulations and scalable, small-scale manufacturing strategies can overcome delivery and production challenges and accelerate the clinical translation of personalized cancer vaccines.

Unlocking The Next Wave Of Cancer Vaccines
Discover how advanced ionizable LNP formulations and scalable manufacturing strategies can accelerate the development and clinical translation of personalized cancer vaccines.

Where Sourcing Meets Science: Smarter Collaboration, Better Outcomes
See how collaboration-driven raw material control reduces variability, improves cell culture performance, and delivers measurable cost and productivity gains in biotherapeutic manufacturing.

OneTeam™: Setting A New Standard for Biotech Program Visibility
An on‑demand session showing how integrated governance and real‑time data boost visibility, collaboration, and control in outsourced gene therapy development.

Industrializing Cell Therapy With Perfusion
Explore how intensified perfusion strategies enable rapid, high‑density CAR‑T expansion, predictive scale‑down modeling, and efficient automated harvest, supporting shorter timelines and higher yields.
Defined Media, Defined Results: Scaling E. coli From Bench To Fermenter
Understand the benefits of chemically defined media in E. coli systems, explore real-world plasmid DNA data, and learn practical strategies for scaling to robust manufacturing.

The Phase 2 Endpoint Dilemma
Phase 2 neuro endpoints can miss subtle change. Learn when composites help or hinder, how to align cognition, function, and biomarkers with real-world data for go/no-go decisions.
Digital Design Meets Proven Performance: The Future Of Single-Use Assemblies
Single-use assembly design is evolving fast. Learn how scalable platforms, digital automation tools, and global supply networks are converging to support sustainable biopharma manufacturing at scale.

Applying AI And Rapid Prototyping To Media And Process Development
Hear from industry experts on how teams can integrate manufacturability considerations earlier through rapid prototyping, data-driven media development, and predictive modeling.

2026 Manufacturing Trends Reshaping Life Sciences
Explore the manufacturing trends shaping 2026, from AI in validated environments to connected systems and digital workforce readiness that are building more agile, resilient, and compliant operations.

Enhanced Biotherapeutic Protein Expression Using Advanced Vector Systems
Learn how optimized expression vector design and data-driven strategies can significantly enhance product titre, quality, and long-term gene expression stability in GS-CHO cell-based manufacturing.
Translating Stem Cell Programs To GMP
A detailed exploration of challenges in advancing stem cell programs to GMP, highlighting strategies to control variability, strengthen process design, and support reliable clinical‑stage manufacturing.
The Future Of CGT: Integrating Digitalization With Regulatory Readiness
Explore how cell and gene therapy manufacturers are scaling from batch‑of‑one to commercial supply while staying compliant, digital, and efficient, as well as strategies to strengthen quality.

Four Common Pitfalls To Avoid In UF/DF Setup And Scale-Up
Learn how small UF/DF decisions can create major scale-up challenges, as well as practical ways to improve membrane selection, reduce manual operations, and boost PD–MSAT alignment.

Bridging Discovery And CMC With Rapid Pools
Learn how early stable CHO expression data reveals manufacturability issues in complex antibodies that transient screening misses, with case studies showing how this changes lead selection.

Charting The Path To First-In-Human: Strategic Readiness For Early Clinical Success
Gain a comprehensive, end-to-end perspective on the journey to first-in-human and understand how early decisions influence downstream success.

Selecting A Prefillable Syringe System With Confidence
Discover an advancement in the prefillable syringe market, uniquely integrating the syringe barrel, plunger, and needle shield/tip cap into a fully harmonized, verified system from a single supplier.
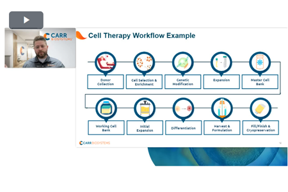
Simplified Harvest And Media Exchange In Cell Therapy
Scale-up often introduces shear stress and process variability. Learn how low-shear, single-use technology stabilizes media exchange and harvest, ensuring consistent cell quality and predictable performance.

Next-Level Cell Lines: An Integrated Approach To Biologic Innovation
See how AI-driven tools and optimized platforms are accelerating biologics development, delivering high-titer clones in weeks, and paving the way for integrated design of cell lines and genetic systems.

Five Practical Considerations To Move From Concept To Clinic
Learn more about five key factors for advancing targeted in vivo LNP programs, from formulation and targeting strategies to bioanalytical readiness and scalability.

Overcoming Regulatory Hurdles In AAV Production
Demonstrating residual reagent clearance is a critical regulatory requirement in AAV manufacturing. Explore how specialized assays and strategic partnerships streamline compliance.

From Lab To Commercialization: Simplifying Bioprocess Scale-Up
Explore how unique cuboid geometries and advanced mixing dynamics create consistent performance from the bench to commercial manufacturing, ensuring flexibility and improved productivity.

CRO Selection Science: Insights From $2B+ Vendor Choices
Discover how data-driven strategies and structured RFPs can simplify CRO selection, improve transparency, and ensure the right operational fit. Learn practical steps to make smarter vendor decisions.
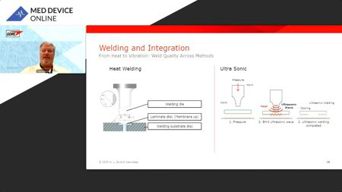
Seal The Deal: Mastering Vent Integration In Medical Devices
Learn how precise vent placement and microfiltration design optimize performance in life science devices. See practical troubleshooting strategies and a real-world case study.

Lessons From FDA 483s And Warning Letters: Cleanroom Compliance
Learn critical lessons from FDA 483 observations and Warning Letters to proactively address common GMP cleanroom compliance failures and build an inspection-ready facility.
Robust And Cost-Effective mRNA Capping
Master the essential strategies for cost-effective and robust mRNA capping at industrial scale. Learn how to successfully evaluate new materials and optimize your synthesis process for maximum efficiency and yield.

Eliminate Risk From Your Viral Vector Tech Transfers
Explore strategies to simplify viral vector tech transfers, reduce risk, and maintain quality under tight timelines, as well as a case study that demonstrates how to streamline this critical process.

Navigating Innovation And Challenges In CGT Bioprocessing
This expert panel breaks down the practical realities of today’s supply chain while forecasting how emerging technologies will redefine quality standards.
