
CELL AND GENE THERAPY ANALYTICS

Analytical Solutions To Help Accelerate Bioprocessing Success
Unlock the power of data-driven bioprocessing. Discover how advanced analytics can optimize your cell culture process, improve performance, and accelerate success.

Rapid Process Development And Technical Support For AAV Scaleup
Accelerate your AAV production journey. Learn how rapid process development and expert support can streamline your path from vial to purified bulk, ensuring scalable and efficient manufacturing.
Maximize Quality Assurance Through Rapid Sterility Testing
This webinar explores mitigating the risk of product loss, and production delays, as well as the critical role rapid sterility testing plays in ensuring the quality and safety of cell therapy products.

Rapid Micro Methods In QC Micro Testing: A NIBRT Perspective
Learn about some of the advanced Rapid Micro Method systems that are being adopted by biopharma companies worldwide, and how they are changing the face of QC Microbiological testing.

Inside Bioprocessing: Multi-Omics
Dive into multi-omics analysis with Paul Gulde, Chengjian Tu, and Femi Egbebi. In biopharmaceutical production, meeting the unique nutrient requirements of the cells is central to achieving optimal results.

An All-In-One Solution For Residual DNA Quantitation
Explore an all-in-one solution for residual DNA quantitation with a 3D lab tour with virtual demos, videos, and interactive instrument guides to experience the full workflow.

Digital Solutions For Accelerated Innovation
In this webinar, hear how Applied Biosystems AccuSEQ software serves as an advanced digital tool that can help streamline bioanalytical processes and accelerate the delivery of life-changing therapies.

Multi-Omics And Bioinformatics In Cell Culture Media Design
Learn about the impact of a multi-omics analysis, utilizing proteomics and metabolomics, in the development and optimization of media with the goal of improving titer, and making development and manufacturing more efficient.

Cell And Gene Therapy Innovations
This interview with a pharmaceutical analytics expert discusses key insights into advances in cell and gene therapy and the strategies used to support customers in improving their therapeutic development.

Validation Of A qPCR Assay For Host Cell DNA Quantitation
Here, we share the development and validation of a new, highly sensitive and accurate integrated solution for detection and quantitation of host cell DNA to help meet regulatory requirements.

Advancing Vaccine Development With Novel Chromatography Solutions & Quality Testing
Learn more on novel chromatography solutions that can help improve the downstream processes of different vaccine types such as mRNA, recombinant proteins, and Virus-Like Particles (VLPs).

Next-Gen Disease Models And Cell Therapeutics: Innovations In AI, Omics And Collaboration
Learn how to leverage AI, omics integration, and collaborative research to build more physiologically relevant disease models and accelerate the development of next-gen cell therapies.

Analytical Approaches For Quantifying Residual Host Cell DNA
In this webinar, we’ll discuss the challenges, risks and considerations involved in developing and using in-house residual testing solutions, and the benefits of using commercial kits for residual DNA testing.
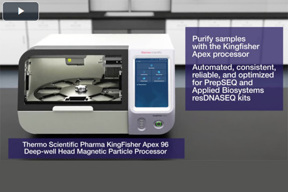
ResDNASEQ Workflow Solution
Explore the Applied Biosystems resDNASEQ workflow solutions. Starting with the sample preparation kit, to purification, quantitating residual host-cell DNA, and analyzing resDNASEQ assays.

Microbial Identification Via DNA-Seq
Learn how DNA sequencing-based microbial identification supports regulatory compliance and enhances contamination control in critical environments.

Key Considerations For Rapid Microbial Methods For Mycoplasma Detection
Learn the importance of demonstrating product specific suitability, defining representative sample, consistent sampling protocols, accommodating volume and turnaround time constraints, and the need to establish equivalence.
Implementing Rapid Microbial Identification In Biotherapy Manufacutring
Learn about rapid microbial identification strategies that enhance environmental monitoring and compliance with regulatory requirements for your manufacturing processes.

Quality Control Assays
Two company leaders detail not only the vectors their respective companies use and why but also the quality control assays performed on viral vectors.

Applying AI And Rapid Prototyping To Media And Process Development
Hear from industry experts on how teams can integrate manufacturability considerations earlier through rapid prototyping, data-driven media development, and predictive modeling.

Simple In-House Mycoplasma Testing Method For Regulatory Expectations And Rapid, Confident, And Actionable Results
In this webinar, we present an overview of worldwide regulatory guidance for mycoplasma testing, as well as a mycoplasma testing method that has been accepted by regulatory authorities worldwide across various therapeutic modalities.

Multi-Omics In Cell Culture Media Design
Discussing the evolution of medium design for cell culture bioprocessing, and the role that multi-omics and bioinformatics can play in improved medium development.

Explore The Analytics Knowledge Hub
Discover smarter solutions with the new Analytics Knowledge Hub that provides articles, webinars, e-books, and infographics designed to enhance and streamline your bioprocess workflow.

Interview With Nico Chow And Sandi True, Field Applications Specialists
Hear Nico Chow and Sandi True, Field Applications Specialists, discuss working with customers to help them evaluate and implement Residual DNA Quantitation, Mycoplasma Detection, and Microbial Identification Assays.
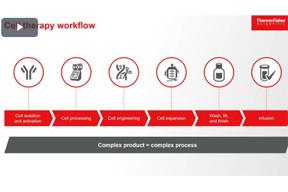
Leveraging Rapid Sterility Testing To Advance Cell Therapy Production
Explore the crucial role of rapid sterility testing in cell therapy manufacturing. This presentation delves into the benefits of swift, accurate detection for product quality and patient safety.

BPI Interviews Purification And Pharma Analytics Leader
Jean Luo, VP of Purification and Pharma Analytics, discusses innovations in bioprocessing, including the development of dPCR assays, lentiviral titer systems, chromatography resins, and supply chain.

Innovative Contamination Control: Enabling Integrity And Efficiency
An integrated approach can enhance contamination control, boost process efficiency, and ensure the production of high-quality cell therapy products.

Leveraging dPCR r Techniques To Quantitate Lentivirus Particles
Discover how digital PCR can streamline lentiviral vector characterization to reduce variability and improve analytical sensitivity. Learn how this approach can help you comply with regulatory guidance.

Introducing The Applied Biosystems Resdnaseq Quantitative DNA System
Thermo Fisher Scientific's portfolio of resDNASEQ solutions is here to meet your specific needs enabling you to test for residual host cell DNA and release your biotherapeutics with confidence in safety and confidence in product quality.

A Straightforward Path Toward Regulatory Compliance, Data Integrity, And Computer Systems Validation
Learn practical strategies for validating microbial identification systems in cGMP. Understand regulatory guidance and master a complete Computer Systems Validation plan for full compliance.

Regulatory Compliance And Viral Vectors
Hear from experts who detail the specific regulatory challenges for advanced therapies, as well as how the regulatory requirements differ in early research and development in academia and in industrial vector production.
