ABOUT ALDEVRON
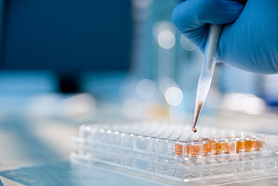
For 25 years Aldevron has been a catalyst, advancing therapeutic and vaccine development through expert plasmid DNA, RNA, and protein custom manufacturing, supporting our clients from discovery to commercialization. Our custom development and manufacturing (CDMO) services have given scientists around the world essential components to accelerate their research and make a difference.
Whether you’re working on a gene therapy, cell therapy, or vaccine development, it starts with a high-quality DNA, RNA, or Protein. You’ll want a partner that can support you from small-scale, pre-clinical work through clinical trials, and eventually at commercial scale with phase-appropriate quality grades, analytics, and regulatory support.
Our current 75,000 square-foot, GMP manufacturing facility has been producing the highest quality plasmid DNA since opening in 2018, to help meet worldwide demand. Our 189,000 square-foot expansion features industry-leading innovations and significantly increases production. This manufacturing facility was visited by the Center for Biologics Evaluation & Research (CBER) in November 2021. CBER conducted a Pre-License Inspection (PLI) and a general GMP audit of Aldevron, LLC. No Form FDA 483 was issued, and the inspection was classified as no action indicated (NAI).
It's our mission to provide products and services that make meaningful contributions to cell and gene therapy. We seek to be the partner of choice with custom manufacturing and/or standardized, catalog products, such as CRISPR nucleases, that support of our clients' comprehensive objectives.
NEXT GENERATION PLASMID TECHNOLOGY
The Power of a Small Backbone for Gene & Cell Therapies
Looking to enhance performance and manufacturing from your DNA backbone, without the regulatory concerns? The small Nanoplasmid backbone fulfills all the requirements. The Nanoplasmid Vector System (<500 bp backbone) provides advantages over existing legacy plasmid DNA backbones and minicircles:
- High Transgene Expression
- Increase Manufacturing Yields
- Improve Safety Profile by Eliminating Antibiotic Resistant Gene
- Save Time and Cost-Effective
Combining characteristics of reduced size and the RNA-OUT non-antibiotic resistance selection marker, the platform is ideally suited for a range of therapeutic applications. Aldevron can provide customized Nanoplasmid DNA for each client.
Webinar 1: Advanced Plasmid Technology: Improved Safety And Performance
Webinar 2: Can A Small Plasmid Produce Huge Benefits? The Power Of Small
Webinar 3: Genome Editing Tools: Beyond Discovery
NEXT GENERATION PLASMID TECHNOLOGY RESOURCES
RNA RESOURCES
FEATURED BROCHURES
CONTACT INFORMATION
Aldevron
4055 41st Avenue South
Fargo, ND 58104
UNITED STATES
NEWS
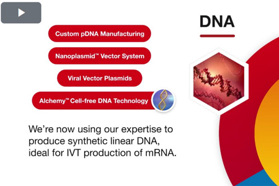
- Aldevron Unveils Next-Generation Alchemy™ Cell-Free DNA Technology For Research Grade Applications
- Aldevron Announces Expansion Of mRNA Production Capability
- Aldevron Launches Eureca-V™ MAD7®, A Type-V CRISPR Nuclease
- Aldevron Licenses The Manufacturing Of A TYPE-V CRISPR Nuclease From Inscripta
- Aldevron Acquires Nature Technology Corporation
- Aldevron Now Offering Its pALD-Help Plasmid For Viral Vector Production In GMP-Source® Quality Level
- In The News: Moderna And Aldevron Announce Expanded Partnership For mRNA Vaccine And Therapeutic Pipeline