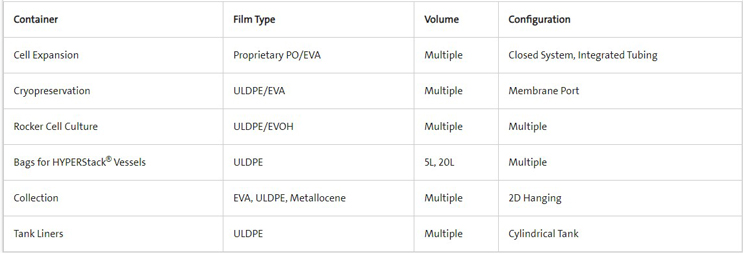
Corning single-use containers are fabricated using single or multilayer films selected for specific bioprocess applications. The films are made using an animal-free, cGMP, ISO 13485 manufacturing process to provide superior strength, biocompatibility, and chemical compatibility.
BioavailabilityOur film for single-use containers undergo a range of biocompatibility tests to ensure there are no adverse effects on any biological material including USP <88> Class VI for in vivoreaction to the material, USP <87> for in vitro reactions (toxicity), USP <661> for extractables, and USP <85> for bacterial endotoxins.