
SUPPLY CHAIN

A Dedicated Team To Safely Store Your Cell And Virus Banks
The safety of your cell and virus banks must be your top priority. Learn about a team dedicated to the safety of your material along with their related services.

BPI Interviews Purification And Pharma Analytics Leader
Jean Luo, VP of Purification and Pharma Analytics, discusses innovations in bioprocessing, including the development of dPCR assays, lentiviral titer systems, chromatography resins, and supply chain.

Bridging The Gap Between Lab Development And GMP Manufacturing
In this first segment of Cell & Gene Live, Optimizing Storage Solutions for iPSCs, expert panelists Pratik Jaluria, Ph.D., SVP Technical Development Strategy at BlueRock Therapeutics, Bruno Marques, Ph.D. VP, Process and Product Development at Century Therapeutics, an...

Balancing Innovation With Manufacturability When GMP-Grade Is Unavailable
Some raw materials simply aren't available in GMP. Experts Yan Zhi and Lawrence Thompson describe risk assessment approaches that inform sourcing decisions in this segment from Cell & Gene Live "Viability On The Line." Some investigational therapies requ...

Singapore Single-Use Site Capacity Expansion
Thermo Fisher Scientific is expanding our single-use manufacturing network globally, an expansion site closer to Asia Pacific region
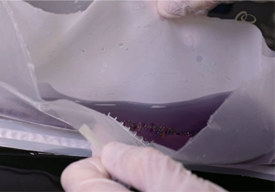
Microcarrier Separation Systems
Ross Acucena, applications director, and Donnie Beers, product manager, introduce our highly functional and adaptable microcarrier cell separation bags designed for single-use processes.

Biopharma Supply, Reimagined: Blarney, A Climate-Neutral Facility For Best-In-Class Filters
Explore how a new, climate-neutral manufacturing facility strengthens biopharma supply chains. Understand the benefits of its advanced filtration technologies and commitment to environmental sustainability.

An Inside Look At Umoja Biopharma And Bristol Myers Squibb's Cold Chains
Umoja's Co-Founder and CTO Ryan Crisman details The CLIMB and how an optimized cold chain is critical in the work being done there. Rookie Gleich, Global Head, Cell Therapy Logistics and Shipping Systems, BMS explains some of the cell therapy temperature standardizati...

Breaking Down Shipping
Here, Umoja's Co-Founder and CTO Ryan Crisman and BMS’ Global Head, Cell Therapy Logistics and Shipping Systems, Rookie Gleich talk about whether temperature monitoring devices are really truly necessary when using a shipper that has been tested and validated to...
Bioprocessing Solutions For Cell Culture
Whatever the scope of your project, you can choose from our unmatched portfolio of high-impact produCTS and services to deliver consistent, scalable solutions.

mRNA For Cancer Immunotherapy 2024 Outlook | Audience Q&A
During our Cell & Gene Live, mRNA For Cancer Immunotherapy 2024 Outlook, our expert panelists answered audience questions regarding patient safety, foreseeable challenges in mRNA cancer immunotherapy for solid tumors, and much more.

Overcoming Cryopreservation Challenges
In this segment, Umoja's Co-Founder and CTO Ryan Crisman and BMS’ Global Head, Cell Therapy Logistics and Shipping Systems, Rookie Gleich share how they are tackling some of the biggest issues in cryopreservation as well as best practices given the complex struc...

Perfusion: Planning A Run
Learn about a logical approach for defining the operation of a perfusion run. We will break down the process using the three following steps: knowledge space, design space, and control space.

Thermo Fisher Scientific Single-Use Global Manufacturing Network
We have implemented a multi-tier strategy to improve the resiliency of our supply chain and created the largest networked single-use manufacturing organization in the world.

Storing Your Banks With Ease And Confidence
Discover a biorepository that will fulfill your need of long-term and short-term storage. You can never be too careful with your cell or virus banks. Explore three global sites that will ensure the safety of your bank.

Supporting The Global Biopharma Industry With Single-Use Capacity Expansion
Explore Cytiva’s brand new single-use production facility in Wales, UK. This facility forms part of Cytiva’s >$1.5 billion investment in increased capacity to support the growing needs of the biopharmaceutical industry and to increase regional supply.

Comparing Manual Vs. Automated Processing And Thawing Systems
In this segment of our Cell & Gene Live, Optimizing Storage Solutions for iPSCs, expert panelists Pratik Jaluria, Ph.D., SVP Technical Development Strategy at BlueRock Therapeutics, Bruno Marques, Ph.D. VP, Process and Product Development at Century Therapeutics, and ...

Regulatory Considerations For An Optimized Cold Chain
Here, Umoja's Co-Founder and CTO Ryan Crisman and BMS’ Global Head, Cell Therapy Logistics and Shipping Systems, Rookie Gleich detail some of the cold chain for cell therapies is beholden to regulatory requirements.

Risk Mitigation Strategies
In this segment, Umoja's Co-Founder and CTO Ryan Crisman and BMS’ Global Head, Cell Therapy Logistics and Shipping Systems, Rookie Gleich explain some of the key risk management mitigation issues regarding managing temperature before...

Supply Chain And Operational Challenges Facing mRNA
Explore some of the biggest supply chain challenges facing mRNA cancer therapeutics and what short-term solutions may exist, starting with Chain of Custody/Chain of Identity.

Optimizing Your Cold Chain For Cell Therapies | Audience Questions
In this segment, Umoja's Co-Founder and CTO Ryan Crisman and BMS’ Global Head, Cell Therapy Logistics and Shipping Systems, Rookie Gleich answer questions from the audience, including the best way to de-risk contract couriers' use of contract carriers for sp...

Inventory Management, Staff Training, And Timeline Optimization
In this segment, Umoja's Co-Founder and CTO Ryan Crisman and BMS’ Global Head, Cell Therapy Logistics and Shipping Systems, Rookie Gleich explain the improvements that can be made in each of the following: inventory management, staff training, and timeline.
...
Robust And Consistent Frozen Storage And Shipment
Learn how a reliable cold chain operation requires the right commercial scale solution, from controlled bag film production to a unified process including reliable control mechanisms.
