
REGULATORY GUIDANCE AND ENABLING COMPLIANCE

Dr. Peter Marks on the Adoption of Novel Precision Analytics
Hear advice for developers who are worried that the FDA will not accept changes in their process that include a new analytical method.
Maximize Quality Assurance Through Rapid Sterility Testing
This webinar explores mitigating the risk of product loss, and production delays, as well as the critical role rapid sterility testing plays in ensuring the quality and safety of cell therapy products.

Rapid Micro Methods In QC Micro Testing: A NIBRT Perspective
Learn about some of the advanced Rapid Micro Method systems that are being adopted by biopharma companies worldwide, and how they are changing the face of QC Microbiological testing.

Understanding AI And Machine Learning's Impact On CGT with Dr. Peter Marks and Dr. Nicole Verdun
During this segment, Dr. Peter Marks and Dr. Nicole Verdun share their take on how AI and machine learning will impact FDA regulation in 2024.

An All-In-One Solution For Residual DNA Quantitation
Explore an all-in-one solution for residual DNA quantitation with a 3D lab tour with virtual demos, videos, and interactive instrument guides to experience the full workflow.

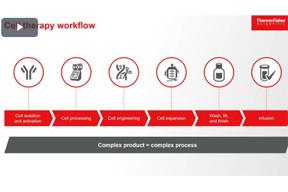
Defining The CMC / Clinical Relationship For Allogeneic Cell Therapies
During this segment, Nguyen and Wagner define why the relationship between CMC and Clinical is so critical, and they also explain what a mutually beneficial partnership between CMC and Clinical looks like.

2025 CGT Regulatory Outlook | Audience Q&A
Kimberly Benton, Ph.D., Master Principal and Head of Regulatory at Dark Horse Consulting and Bambi Grilley, RPh, RAC, CIP, CCRC, CCRP, Director, Clinical Research and Early Product Development Center for Cell and Gene Therapy, Professor of Pediatrics Baylor College of Med...

Cell And Gene Therapy Innovations
This interview with a pharmaceutical analytics expert discusses key insights into advances in cell and gene therapy and the strategies used to support customers in improving their therapeutic development.

Tackling Cell And Gene Therapies' Top 3 Regulatory Impediments: Audience Q&A
During each Cell & Gene Live, our audience has the opportunity to submit real-time questions for our expert panelists. Watch and listen as Nina Hunter, Ph.D., VP Corporate Strategy at REGENXBIO and Adora Ndu, PharmD, JD, Chief Regulatory Affairs Officer at BridgeBio p...

Validation Of A qPCR Assay For Host Cell DNA Quantitation
Here, we share the development and validation of a new, highly sensitive and accurate integrated solution for detection and quantitation of host cell DNA to help meet regulatory requirements.

Advancing Vaccine Development With Novel Chromatography Solutions & Quality Testing
Learn more on novel chromatography solutions that can help improve the downstream processes of different vaccine types such as mRNA, recombinant proteins, and Virus-Like Particles (VLPs).

Challenge #1: The Changing Regulatory Landscape
By 2030, 60 CGTs are expected to reach regulatory approval. One of the biopharmaceutical industry's most closely watched regulatory developments is the FDA’s reauthorization of the Prescription Drug User Fee Act (PDUFA) or PDUFA VII, as it provides resources for...
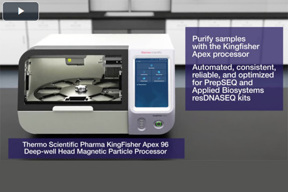
ResDNASEQ Workflow Solution
Explore the Applied Biosystems resDNASEQ workflow solutions. Starting with the sample preparation kit, to purification, quantitating residual host-cell DNA, and analyzing resDNASEQ assays.

Microbial Identification Via DNA-Seq
Learn how DNA sequencing-based microbial identification supports regulatory compliance and enhances contamination control in critical environments.

Key Regulatory Challenges, Innovations, And Opportunities For CGT In 2025
Kimberly Benton, Ph.D., Master Principal and Head of Regulatory at Dark Horse Consulting and Bambi Grilley, RPh, RAC, CIP, CCRC, CCRP, Director, Clinical Research and Early Product Development Center for Cell and Gene Therapy, Professor of Pediatrics Baylor College of Med...

How Past Gene Therapy Approvals Are Shaping the Future with Dr. Peter Marks
In this segment of Cell & Gene Live, Dr. Peter Marks shared what he and his team have learned from current gene therapy approvals to accelerate future approvals.

Key Considerations For Rapid Microbial Methods For Mycoplasma Detection
Learn the importance of demonstrating product specific suitability, defining representative sample, consistent sampling protocols, accommodating volume and turnaround time constraints, and the need to establish equivalence.

Why Clinical Requirements Change: Dr. Peter Marks on Pre-IND Meeting Pathways
Learn how clinical requirements change after the FDA and a company agree on a pathway during a pre-IND meeting and how the company might recover as a result.
Implementing Rapid Microbial Identification In Biotherapy Manufacutring
Learn about rapid microbial identification strategies that enhance environmental monitoring and compliance with regulatory requirements for your manufacturing processes.

CMC Comparability: Transitioning to Commercial Manufacturing with Dr. Nicole Verdun
In this segment, Dr. Nicole Verdun shares some of the challenges demonstrating CMC comparability during late-stage product development (i.e., pivotal or Phase 3 clinical trial) that occur while transitioning to commercial manufacturing.

Focal Points for 2024: Dr. Verdun and Dr. Marks Talk Regulatory Challenges for Cell-Based Therapies
Dr. Verdun and Dr. Marks provide detailed feedback on the regulatory challenges facing cell therapy and explain the Agency’s most important focal points for 2024, patient accessibility, and more.

Simple In-House Mycoplasma Testing Method For Regulatory Expectations And Rapid, Confident, And Actionable Results
In this webinar, we present an overview of worldwide regulatory guidance for mycoplasma testing, as well as a mycoplasma testing method that has been accepted by regulatory authorities worldwide across various therapeutic modalities.

How The FDA's 2024 Draft Guidances Will Influence CGT Regulation In 2025
Kimberly Benton, Ph.D., Master Principal and Head of Regulatory at Dark Horse Consulting and Bambi Grilley, RPh, RAC, CIP, CCRC, CCRP, Director, Clinical Research and Early Product Development Center for Cell and Gene Therapy, Professor of Pediatrics Baylor College of Med...

mRNA For Cancer Immunotherapy 2024 Outlook | Audience Q&A
During our Cell & Gene Live, mRNA For Cancer Immunotherapy 2024 Outlook, our expert panelists answered audience questions regarding patient safety, foreseeable challenges in mRNA cancer immunotherapy for solid tumors, and much more.

Explore The Analytics Knowledge Hub
Discover smarter solutions with the new Analytics Knowledge Hub that provides articles, webinars, e-books, and infographics designed to enhance and streamline your bioprocess workflow.

Interview With Nico Chow And Sandi True, Field Applications Specialists
Hear Nico Chow and Sandi True, Field Applications Specialists, discuss working with customers to help them evaluate and implement Residual DNA Quantitation, Mycoplasma Detection, and Microbial Identification Assays.

Collaborating with the FDA: Dr. Peter Marks and Dr. Nicole Verdun Share Advice For Small And Emerging Biotechs
In this segment, Dr. Peter Marks and Dr. Nicole Verdun share their advice and best practices for small and emerging biotechs when working with the FDA.
Leveraging Rapid Sterility Testing To Advance Cell Therapy Production
Explore the crucial role of rapid sterility testing in cell therapy manufacturing. This presentation delves into the benefits of swift, accurate detection for product quality and patient safety.

Challenge #2: Rare Or Ultra-Rare Study Design Considerations And Use Of Existing Data
There are more than 7,000 unique, rare diseases recognized in the United States, and 300 million people worldwide living with a rare disease; patients often experience a long journey to receive their diagnosis. Our expert panelists, Nina Hunter, Ph.D., VP Corporate Strate...

Navigating Global Regulatory Landscapes In An Evolving Biopharma Industry
Minimize regulatory risks and ensure compliance in the evolving biopharma sector. Watch this on-demand discussion for insights on change management, supplier collaboration, and future market trends.
