Process Engineering's Key Role In Sterile Injectable Facility Design
By Sanjeev Kumar, NIRAS A/S, Denmark

Sterile injectable medicinal products represent one of the most demanding areas of pharmaceutical manufacturing. They include vaccines, insulin, oncology drugs, antibiotics, advanced biologics, monoclonal antibodies, blood plasma-derived products, and emerging cell and gene therapies — all of which require uncompromising sterility and quality.
Because these products are administered directly into the bloodstream or tissue, they bypass the body’s natural defenses and allow no margin for error; therefore, product quality is of the uppermost importance.1,2,3 This drives complex facility design challenges, spanning aseptic processing strategies, contamination control, cleanroom architecture, utilities, automation, and life cycle robustness.
The process engineer — together with operators, quality teams, and subject‑matter experts — understands the end‑to‑end flow, how raw materials become sterile drug product, how exposure risk evolves across each unit operation, and how failures manifest as sterility loss, endotoxin contamination, or data integrity gaps.
Process engineering does not simply support facility design; it defines the technical and regulatory constraints within which all other disciplines must operate.1,2,3,4
By integrating process engineering and infrastructure design, we translate regulatory and product requirements into efficient, scalable, and sustainable sterile injectable facilities. This article explores the key design considerations and challenges behind sterile injectables facility design.
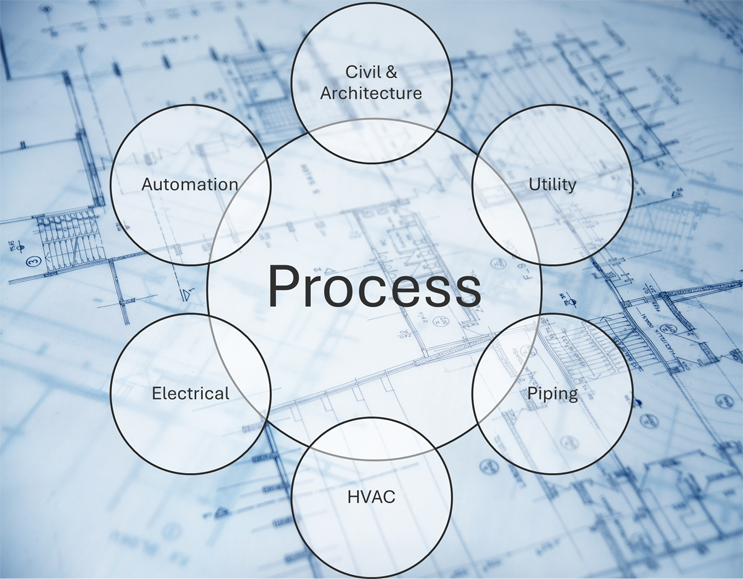
Venn diagram of process engineering within the project
Process Design Engineering: Key Considerations
The process engineer, together with the end user, environmental health and safety, and QA/QC, acts as the lead technical strategist and decision owner, defining the critical process requirements that anchor the entire project.
These requirements — derived from product characteristics, process needs, and operational strategy —directly drive the design of a sterile injectable facility. Each of these factors directly influences facility layout, cleanroom classification, equipment selection, automation strategy, and long-term operational efficiency.
Key considerations include:
- Capacity and throughput
Annual demand stock keeping unit per year, batch size, campaign duration, single product or multiproduct facilities - Format philosophy (single vs. combination line)
Dedicated filling line for vial, syringe, or cartridges or combination line for all formats - Product container type
Vials, syringes, cartridges - Process schedule
Simultaneous operations, peak demands and equipment occupancy - Container preparation strategy
Ready-to-use (prewashed and presterilized) or bulk containers requiring in-house washing, depyrogenation, and sterilization - Final dosage forms
Sterile liquid or freeze/spray-dried products - Product characteristics
Toxic, potent, or biologically active compounds - Contamination control technology
Use of isolators or restricted access barrier systems (open or closed RABS) - Process architecture
Open versus closed systems and the use of single-use versus multi-use equipment - Regulatory and equipment knowledge
Market-specific regulatory requirements and knowledge of filling and drying techniques
- Sterilization strategy
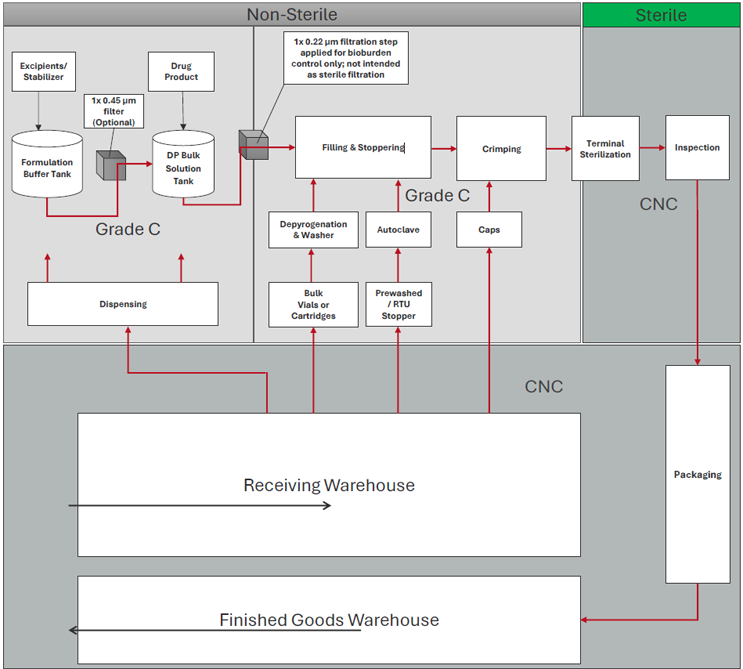
Sterile injectable products are primarily manufactured using one of two approaches: terminal sterilization or aseptic processing.
Terminal sterilization
Terminal sterilization may be performed when products can withstand:
- moist heat sterilization,
- dry heat sterilization,
- sterilization by radiation, or
- sterilization with ethylene oxide based on process requirements
From a regulatory and contamination control perspective, terminal sterilization offers a higher sterility assurance level2,3 and simpler facility design compared to aseptic processing.
Typical process flow: Terminally sterilized process
Aseptic processing
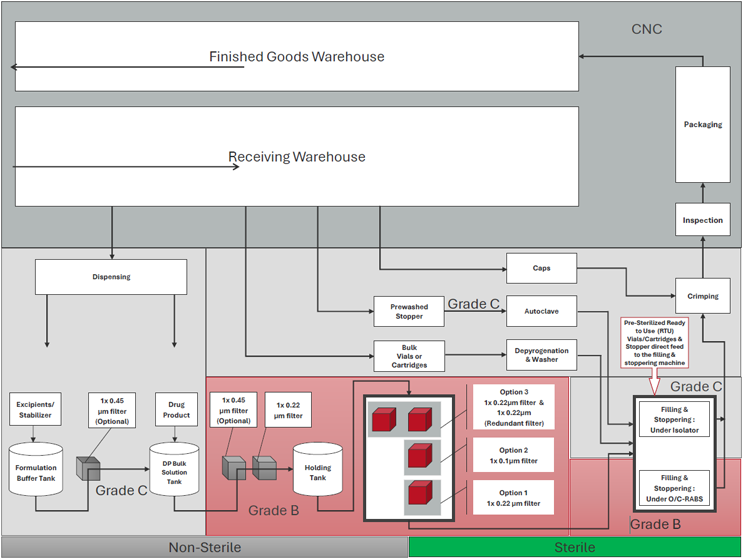
Where terminal sterilization is not feasible — for example, due to product sensitivity to heat or radiation — the product must be aseptically processed.
In aseptic processing, the primary objective is to maintain sterility throughout every stage of manufacturing. Final product solutions are sterilized by filtration through a validated sterile-grade filter, typically with a nominal pore size of ≤0.22 µm, or ≤0.1 µm for higher-risk applications, very small batch sizes, or special cases, under controlled aseptic conditions.
Aseptic processing places significantly higher demands on facility design, environmental control, operator practices, and contamination control strategies.1,2,5
Typical process flow: Aseptic process
Civil And Architectural Design Considerations
Sterile injectable facilities are designed from the inside out,1,4,6 with the process engineer defining the facility concept and setting the boundary conditions for civil and architectural design criteria.
Early involvement enables definition of the process flow philosophy, including vertical, stepped, or horizontal layouts. Decisions related to GMP requirements, material and personnel flows, segregation of clean and non-clean areas, and the location of in-process QC laboratories, control rooms, and interfaces between production and support areas directly influence building dimensions and structural design.
Accurate equipment data — such as dimensions, maintenance clearances, and operational loads — support correct room sizing and structural design, including safe floor loading and vibration control for processing equipment.
Early decisions on utility routing, installation access, and maintenance pathways help minimize operational disruption. Requirements for viewing corridors, goods lifts, and material handling systems must also be defined early to support safe, compliant, and efficient facility operation.
HVAC Design Considerations
Pharmaceutical HVAC design must be based on a thorough understanding of the manufacturing process, product characteristics, and associated operational risks. Clear and early communication from process engineering to HVAC engineering is essential to ensure that product-related and personnel-related risk considerations are correctly translated into a compliant and fit-for-purpose HVAC system design.
Early involvement enables the definition of area classification, material and personnel airlocks, partial air recirculation versus fully exhausted areas, grouping of similar spaces, and other architectural and operational aspects that directly influence HVAC system design.
These inputs have a significant impact on key system parameters, including required air change rates, supply and exhaust airflow quantities, and pressure differential control.
Accurate definition of process-driven inputs — such as area classification, contamination control technologies, room pressure zoning, and heat dissipation from process equipment — supports correct HVAC design.
HVAC systems must therefore be designed for worst-case operational scenarios,1,2,4 ensuring stable environmental conditions during peak occupancy.
Utility Design Considerations
Utility systems for sterile injectable manufacturing must be designed based on a thorough understanding of process requirements, product quality needs, and operational continuity. Utilities play two critical roles: supporting the manufacturing process and directly participating in the process.
Process-contact utilities come into direct contact with the product or product-contact surfaces and therefore have a direct effect on product quality. These include water for injection (WFI), pure steam, and process gases such as nitrogen, argon, and process air. As critical quality utilities, they must be designed, generated, stored, and distributed in full compliance with applicable pharmacopeial standards and requirements to ensure consistent quality, sterility, and system integrity.2,4,7
Process-support utilities do not directly contact the product but are essential for the reliable and controlled operation of manufacturing and support systems. These include instrument air, chilled water, cooling water, hot water, potable water, and plant steam. Although indirect, their availability, quality, and reliability are critical for maintaining stable process conditions and preventing operational disruptions.
Utilities must be assessed on an hourly, daily, and batch basis, considering peak loads, simultaneous operations, cleaning and sanitization cycles, and potential future expansion. These evaluations form the basis for defining the required capacity of utility generation, storage, and distribution systems.
Process-driven utility design is therefore essential — not only to ensure uninterrupted production but to protect long-term compliance, cost efficiency, and operational resilience.
Piping Design Considerations
Process knowledge is a critical input for effective piping design and 3D model development in pharmaceutical projects. A thorough understanding of the process ensures defined flow pathways, proper equipment integration within rooms, and accurate positioning of utility drop points, drain connections, venting provisions, and required safety devices.
Accurate equipment data — including overall dimensions, nozzle sizes, orientations, and the locations of utility and drain connections — must be provided by the process discipline before the piping engineer begins their work.
Piping and tubing specifications, including nominal sizes, end connections, insulation requirements, and materials of construction must be clearly defined and communicated to the piping engineers. These parameters are required to allocate sufficient space for piping, supports, and maintenance access, and to develop effective routing strategies within the building.
Dedicated piping shafts or corridors for process transfer lines, product-contact utilities, and process-support utilities should be identified and planned early in the design phase.
Proper segregation of piping systems is essential1,4,7 to avoid congestion and clashes with other building services — such as electrical cable trays and HVAC ductwork — which are frequently encountered during project execution.
Electrical Design Considerations
Electrical systems in pharmaceutical manufacturing facilities are designed to supply reliable power to process equipment, utilities, and support systems essential for continuous and compliant operations.
Total connected load and maximum peak demand are key drivers of facility electrical design. Process knowledge of operating modes, duty cycles, and simultaneity is required to accurately define connected, operational, and peak loads, which directly influence system sizing and configuration.
Backup power systems — such as diesel generators and uninterruptible power supply (UPS) systems — are essential for critical process and support equipment. The scope and duration of backup power must be defined based on process criticality.
In hazardous areas, process equipment and associated electrical components must be correctly classified and selected as explosion-proof or intrinsically safe in accordance with applicable standards and area classification requirements.1,4
Finally, the location and segregation of control panels, electrical panels, switchboards, and UPS systems must be agreed upon early through coordination between process, architecture and electrical engineers to ensure a safe, maintainable, and compliant design.
Automation Design Considerations
A clear understanding of the manufacturing process is essential to define control strategies, system boundaries, and automation logic that directly affect sterility assurance, product quality, and GMP compliance.
For process equipment, the process engineer ensures that process intent, critical parameters, and regulatory requirements are accurately translated to original equipment manufacturers (OEMs), enabling automation that correctly executes the manufacturing process.
At the control system level — including PLC, DCS, and SCADA — process knowledge defines what must be controlled, monitored, or interlocked under normal and abnormal operating conditions. Setpoints, alarms, sequences, and fail-safe behavior must be defined so automation systems actively protect sterility, product quality, and data integrity, rather than merely operate equipment.
Process engineers also support the design of environmental monitoring systems, building management systems, and automated material handling by aligning functionality with process exposure, contamination risk, and material flow.
At the enterprise and execution levels, process engineers provide essential inputs to enterprise resource planning (ERP) and manufacturing execution system (MES) systems — batch recipes, sequencing, operator instructions, in-process controls, hold points, and electronic records — ensuring consistent execution, data integrity, and full traceability across the manufacturing life cycle.1,2,4,7
Process Equipment Design Considerations
Process knowledge should extend beyond core process design to include a strong understanding of equipment design and associated technologies. The process engineer plays a key role in evaluating and selecting the right technology, working closely with original equipment manufacturer (OEMs) to align equipment design with product and process requirements. Early and continuous OEM involvement enables optimized equipment design, effective process room design, well-defined utility drop points, seamless integration with upstream and downstream equipment, regulatory compliance, and future-ready facilities.1,2,3,4,5,6,7,8
Summary
Every project is unique and cannot follow a single design approach. Process knowledge acts as the project’s brain, guiding decisions across conceptual, basic, and detailed design phases. Its true value lies in delivering the right information to the right stakeholders at the right time—transforming uncertainty into informed decision-making. Without strong process engineering leadership, sterile injectable facilities may be built, but they will struggle to operate reliably, compliantly, and economically over their lifecycle.
References:
- ISPE: Baseline Guide Vol 3: Sterile Product Manufacturing Facilities (3rd Edition)
- “Annex 1: Manufacture of Sterile Medicinal Products”
- PIC/S GMP Guide (PE 009 17)
- ISPE: Good Practice Guide: Good Engineering Practice (Second Edition)
- ISPE: Baseline Guide Vol 7: Risk‑Based Manufacture of Pharmaceutical Products
- ISPE: Baseline Guide Vol 6: Biopharmaceutical Manufacturing Facilities (3rd Edition)
- ISPE: Baseline Guide Vol 4: Water & Steam Systems (3rd Edition)
- ISPE: Baseline Guide Vol 5: Commissioning & Qualification (2nd Edition)
About The Author:
 Sanjeev Kumar is a seasoned process engineer with NIRAS and has deep expertise in monoclonal antibody (mAb) production, blood plasma fractionation, and sterile injectable facility design. With a strong foundation in biopharmaceutical manufacturing and equipment life cycle management, he excels in equipment design, operation, troubleshooting, and CQV execution and is known for his ability to coordinate across cross-functional teams and stakeholders. His hands-on experience with bioprocess systems and clean utility operations makes him a trusted contributor to greenfield, retrofit, and complex expansion projects in the biopharmaceutical industry.
Sanjeev Kumar is a seasoned process engineer with NIRAS and has deep expertise in monoclonal antibody (mAb) production, blood plasma fractionation, and sterile injectable facility design. With a strong foundation in biopharmaceutical manufacturing and equipment life cycle management, he excels in equipment design, operation, troubleshooting, and CQV execution and is known for his ability to coordinate across cross-functional teams and stakeholders. His hands-on experience with bioprocess systems and clean utility operations makes him a trusted contributor to greenfield, retrofit, and complex expansion projects in the biopharmaceutical industry.