Navigating GMP Biosafety Challenges In ATMP Manufacturing
By BioPhorum

Advanced therapy medicinal products (ATMPs) — including cell, gene and RNA therapies — are leading the charge in healthcare innovation while introducing new complexities to biopharmaceutical manufacturing. Unlike traditional biologics, ATMPs often use genetically modified organisms (GMOs), replication-competent viruses, and human-derived materials. These components bring unique biosafety risks that may not be adequately addressed by standard facility designs. The integration of biosafety principles into the design of facilities that adhere to GMP is therefore essential, not only for regulatory compliance but also to protect personnel, patients, and the surrounding environment.
We conducted a benchmarking survey that revealed a wide variation in how biosafety is managed within GMP environments, underscoring the clear need for harmonized guidance. This article uses a subset of the survey data and compares companies in the U.S. and EU, showing distinctions between new builds, retrofits, and sites to illustrate key trends and contextual differences. Building on those insights, it sets out a structured, risk-based approach for integrating biosafety into ATMP facilities, emphasizing early involvement of multidisciplinary teams and robust cross-functional collaboration to address the unique challenges these therapies present. The examples provided focus on viral-based ATMPs, rather than synthetic models, to reflect the distinct biosafety considerations associated with viral vectors.
Biosafety For ATMP Facilities
Safeguarding individuals, including facility personnel and patients, from exposure to harmful biological agents is essential for any biopharmaceutical facility. ATMP facilities pose unique challenges and risks, as they often involve complex and novel biological materials, such as GMOs and nontraditional biological components (e.g., human cell lines). These materials have an elevated biosafety risk over traditional biological processes and typically require stringent containment and control measures. Manufacturers bear a corporate and social responsibility to implement robust biosafety measures to fully address these risks.
Figure 1 highlights the differences in biosafety control implementation between retrofit and new ATMP facilities (note: the percentages do not represent the proportion of all companies but a percentage of total respondents from the subset). New facilities most commonly employ structured controls, such as training and system design, reflecting a proactive approach to mitigating worker exposure to biohazardous single-use system failures. In contrast, retrofit facilities demonstrate a more varied approach to control, with no single measure dominating. The higher use of physical containment (e.g., enclosures over points of failure) in a retrofit facility may reflect constraints in modifying existing infrastructure. The data supports the importance of integrating biosafety measures early in facility designs to ensure consistent and effective worker protection.
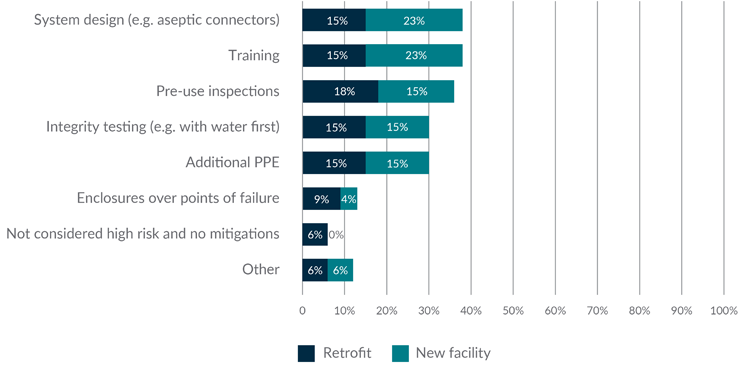
Figure 1: Controls to mitigate worker exposure to biohazardous single-use system failures
Although most organizations report strong leadership and financial support for biosafety in GMP facilities (see Figure 2), many facilities still struggle to balance GMP and biosafety requirements (see Figure 3), particularly in the U.S. To close this gap, stakeholders must make strategic investments to enhance financial support and ensure effective planning and resource allocation for biosafety infrastructure.
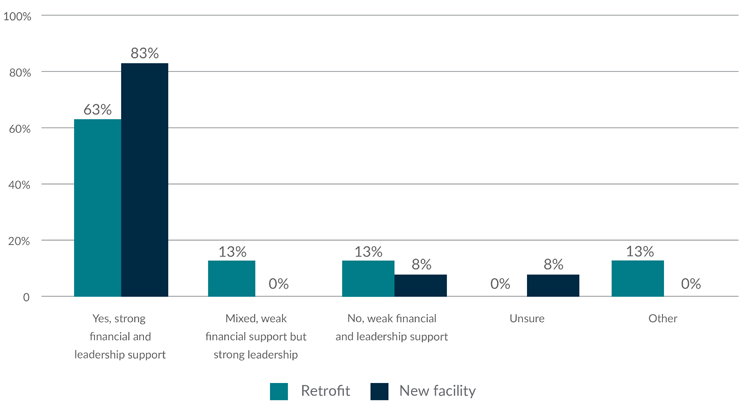
Figure 2: Support for biosafety in GMP facilities
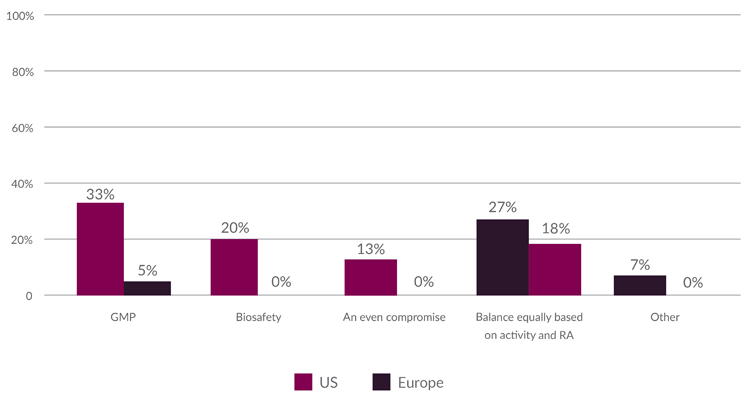
Figure 3: Precedence of GMP or biosafety
Facility Design
As ATMP facilities often operate at the intersection of complex biosafety needs and strict GMP criteria, thoughtful planning and collaboration are paramount to fostering environments that not only support innovative therapies but also ensure operational safety and regulatory alignment. A comprehensive, multidisciplinary risk assessment process is the key to achieving the safe production, handling, and containment of biological materials while also maintaining product integrity and meeting GMP standards. Below are some facility design strategies tailored to address these dual requirements, focusing on unique considerations that are vital for ATMP environments during both standard operations and upset conditions, such as large-scale spills.
Biocontainment Considerations
During routine operations, the chief objectives are to prevent worker exposure, particularly to aerosols, and to contain biological materials to minimize cross-contamination risks and prevent environmental release. When hazards cannot be eliminated or substituted, the most effective mitigation strategy is to implement engineering controls. These may include functionally closed systems validated for containment and equipped with continuous monitors, prefailure alarms, or automatic shutdowns, as well as barrier devices such as biological safety cabinets.
The selection of fit-for-purpose engineering controls requires a comprehensive risk assessment that considers issues such as:
- known and potential biological hazards
- process requirements
- equipment-specific needs (e.g., placement and required services)
- availability of backup power or an uninterruptible power supply.
Safety Systems And Cross-Contamination Control
When determining whether to install safety equipment, such as emergency eyewash stations and safety showers (EWSS), planners must look beyond placement (e.g., the GMP envelope) and address how ATMP-contaminated water from equipment use would be contained and disposed of. The assessment should not only cover biological materials used to produce ATMP products but also corrosive chemicals such as cleaning solutions. EWSS requirements can clash with GMP requirements that prohibit the use of nonsterile water in cleanroom environments, given the need for routine flushing of units and water containment (e.g., floor drains). As an alternative, portable eyewash/drenching stations and/or saline eyewash bottles can be substituted for plumbed units, either to provide flow for the required duration of flushing or as a supplemental unit while personnel are on the way to primary equipment, so long as they are properly managed and replaced before expiration.
Facility Control
As the field of ATMPs continues to advance, achieving a balance between biosafety measures and GMP regulations is crucial for effective facility operational design. Below are the design considerations necessary for achieving this balance, emphasizing the importance of both product and environmental safety in ATMP facilities. The balance between biosafety and GMP requirements guides choices and influences nearly every aspect of facility design, from physical security to personnel protocols. In ATMP facilities, controls must extend beyond GMP and product control to encompass biohazard containment, environmental protection, and biosecurity measures.
Physical Security And Segregation
The foundation of facility security begins with implementing controlled access systems, such as badge readers, to regulate entry into buildings, labs, cleanrooms, and other critical areas. Exterior security must also be considered, including measures such as strategic lighting and surveillance cameras. Regulatory frameworks such as the National Institutes of Health Guidelines and the World Health Organization biosafety guidance provide essential recommendations, with geographical considerations influencing security needs, e.g., EU facilities often require gated access.
Room-specific security measures are critical. Access controls, such as eye scanners for B-grade areas, can be implemented with access linked to personnel training records and qualifications, so that only qualified staff can enter. Surveillance cameras inside secured areas can serve dual purposes, i.e., monitoring safety and supporting risk management. Livestream monitoring can support quality assurance, auditing, and accident investigations, though privacy policies and regional regulations must be addressed, especially regarding audio recording.
Real-Time Monitoring And Equipment Redundancy
Mission-critical equipment, facilities, and utilities require continuous monitoring, with round-the-clock alarm tracking during crucial operations. Rapid response should be enabled through shift services supported by a distributed control system (DCS) that leverages continuous monitoring to ensure malfunctions are detected, addressed, and corrected immediately. An equipment management database should complement the DCS by scheduling and tracking preventive maintenance, calibrations, and requalification activities, ensuring equipment is serviced regularly to avoid unexpected breakdowns. This structured approach enables operators to react to malfunctions immediately, isolate problem areas, and make corrective adjustments before a minor deviation becomes a major failure.
Contamination Control And Facility Services
Compared with general biologics, ATMP facilities require additional considerations, particularly in containment, personnel exposure risk management, and oversight of service providers. Engineers responsible for maintaining high-efficiency particulate air systems may require specialized PPE and biosafety training due to potential exposure risks. External contractors and service providers face the same exposure risks as full-time staff and therefore need to be explicitly informed of potential biohazard or GMO risks and provided with appropriate PPE and safety protocols, such as the use of bag-in-bag-out filter changes. Also, their environment, health, and safety (EHS) programs should be reviewed to ensure they align with the facility’s biosafety standards, reinforcing both contamination control and worker protection.
Personnel Training And Qualification
Workforce training is equally critical for the safe operation of an ATMP facility. Survey findings indicate that, regardless of geographical location or whether the facility is newly built or existing, companies are adopting a blended approach to address diverse learning preferences, operational demands, and site-specific requirements. The absence of a single dominant training method highlights the complexity of GMP ATMP environments, where a balanced mix of theoretical, practical, and safety-focused training is essential to ensure workforce readiness.
Emergency Preparedness, Business Continuity, And Quality Systems
Regular alarm drills in line with EHS/biosafety assessments and post-drill analysis (including feedback and lessons learned sessions) keep emergency teams prepared. To ensure readiness, facilities should conduct periodic decontamination drills that culminate in a lessons learned debrief. To help with rapid response capabilities, it is advisable to consider establishing direct connections with local fire departments, hospitals, and other relevant emergency response organizations. This will facilitate coordination of response plans, the execution of joint drills, and training exercises, ensuring that first responders are well acquainted with the facility layout and procedures.
Occupational Safety
Even the best-designed facilities cannot eliminate every hazard associated with ATMPs. Operators may still face occupational exposure risks while also representing a potential source of contamination. Because ATMPs are relatively new, their complete exposure profile may not yet be known; therefore, a conservative, risk-based approach remains essential. For example, if a person is inadvertently inoculated with a viral vector, the immediate impact depends on various factors, including the nature of the vector and the specific genes delivered. The potential long-term health effects are also uncertain and require further study and monitoring to fully assess ongoing impacts.
Identifying And Characterizing Risks
The first step in evaluating the risks associated with ATMPs is to determine how the agent could enter the body (e.g., through dermal contact or mucous membranes) and then select appropriate control measures and evaluate the remaining risk after the controls have been implemented. Risks arise from the materials involved, such as biological materials, and the processes being conducted. Risk profiles can vary between a drug substance and its drug product, as well as their intermediates, even when they share the same molecule and physical form. The drug substance is the processed product, which is still subject to safety testing, such as tests for adventitious viruses. Conversely, the drug product represents the final stage of the pharmaceutical therapy having been released at the drug substance phase, which may reduce the associated safety concern from BSL-2 to BSL-1.
Identifying Appropriate Safeguards
Once risks have been identified, appropriate controls and practices must be implemented to mitigate them. Gene therapy products are typically classified into risk groups and BSLs. For proteins (e.g., monoclonal antibodies) and small molecules, the general industry approach involves establishing an occupational exposure limit, considering biocontainment requirements, selecting a suitable environment, identifying suitable disinfectants, and selecting appropriate PPE.
Emergency Preparedness And Response Program
When working in a GMP area, emergency preparedness, occupational health considerations, and a response plan must be addressed to ensure employee safety and well-being. Personnel may be exposed to various chemical agents and biological hazards, requiring thorough risk assessments, implementing appropriate safety measures, and establishing a robust training program to address emergency preparedness and response. The best way to ensure consistent adherence to safety controls and prevent unsafe activities is to foster a strong safety culture in the organization. Personnel who understand the “why” and “how” of safety measures are equipped to apply critical thinking to unsafe or questionable scenarios, so that they can spot hazards early and intervene before an incident occurs.
Conclusion
The successful integration of biosafety and GMP within ATMP facility designs is a testament to an organization’s commitment to innovation and safety, driven by robust leadership. Leadership is pivotal in steering these efforts, ensuring the facility design adheres to regulatory requirements while supporting advanced therapeutic processes without compromising safety. This leadership-driven integration enables facilities to fully address and incorporate comprehensive safety measures into ATMP facility designs, ensuring potential risks are considered and mitigated while fostering a robust culture of safety and compliance. By harmonizing GMP with biosafety, organizations establish a clear objective to protect product integrity, safeguard personnel, and ensure environmental safety.
This article summarizes the main points from a recent BioPhorum publication on this topic. To learn more, check out the full paper, Biosafety in the driver’s seat: Navigating GMP challenges in ATMP design. It includes an appendix of common industry challenges and actionable solutions.
