Krakatoa® K500 On-Demand Media And Buffer Manufacturing System
Enable co-location of cell culture media and buffer manufacturing and bioproduction with Krakatoa K500, the first bioreactor-scale pod-based manufacturing system. Produce up to 500 liters of sterile solutions on-demand in less than 3 hours.
Transform Your Media and Buffers for Bioproduction
Lower Acquisition Cost
Powder-filled, single-use pods and on-demand production reduces the number of consumables required, eliminates cold-chain transport, and significantly reduces incoming QC.
Streamline Operations
Automated, closed, single-use workflows with integrated sterile filtration, inline monitoring, and digital batch records remove the need for prep tanks, refrigerated storage, cleaning, and manual documentation.
Shorten Lead Times
Produce 35–500 liters of sterile media or buffers in as little as 3 hours on-site and on-demand, eliminating procurement, shipping, and on-site storage.
Step Into the Future of Bioproduction
Despite advances in upstream and downstream processing, cell culture media and buffer manufacturing has remained largely unchanged relying on manual media and buffer preparation, liquid suppliers, and complex logistics. Krakatoa K500 ushers in a new era: bioreactor-scale sterile media and buffer manufacturing on-demand and at point-of-use within 3 hours.
Proven Functionality
Solubilization
Amino Acid Profile Confirms Solubilization Consistency
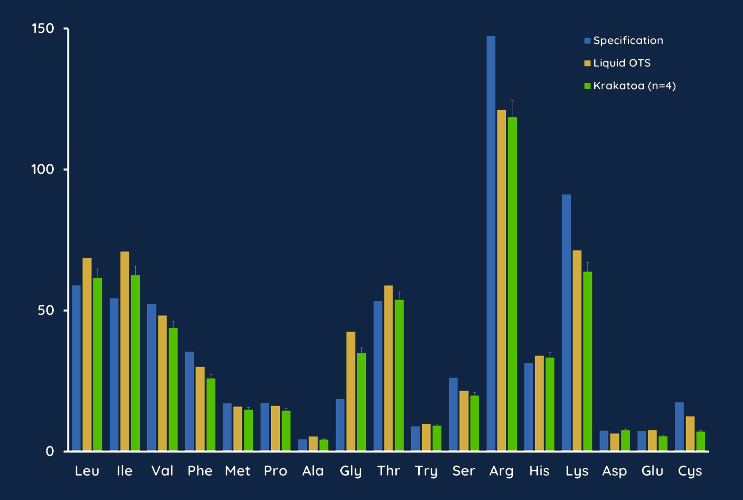
Consistency of your cell media product is critical to the success of your development. Our protocols for pod filling and solubilization ensure media with equivalent quality and solubilized ingredient specifications as off-the-shelf media. In a direct comparison of Krakatoa mixed media and off-the-shelf media (DMEM/F-12 supplemented with 10% FBS), both media showed equivalent levels of amino acids when compared to a control specification.
Proliferation
Krakatoa Media Performs Equivalently to Off-the-Shelf Media
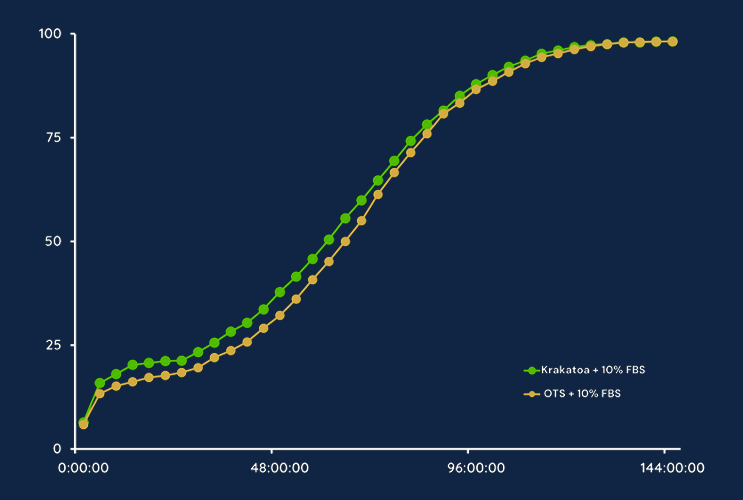
Producing media sustainably is our goal to cure the planet, but producing quality media is a necessity to cure patients. Krakatoa media performs comparably to off-the-shelf media when analyzing cell proliferation and population doubling. In a comparison of off-the-shelf DMEM/F-12 supplemented with 10% FBS vs Krakatoa DMEM/F12 supplemented with 10% FBS on MSCs, our data shows equivalent cell confluence % from Day 0 through Day 6.
Filterability
Krakatoa Can Manufacture Complete Media with Large Proteins
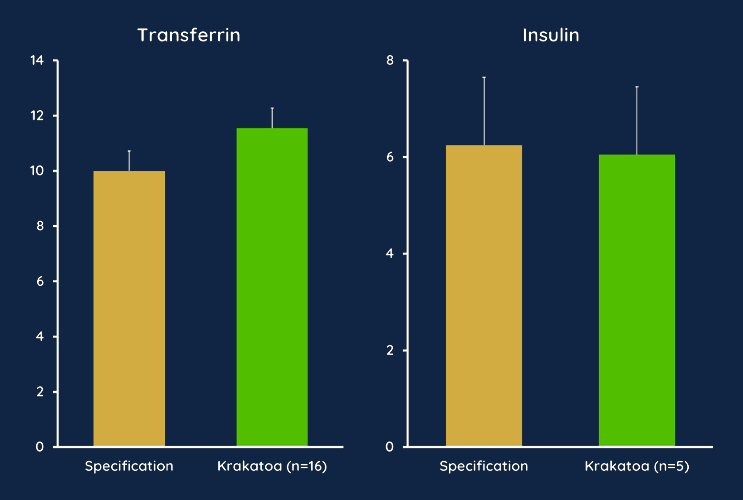
Krakatoa was evaluated for its capability in mixing media supplemented with large proteins. In our study, we manufactured CMRL media, a complex media formulation supplemented with insulin and transferrin. Media was tested for protein content after filtration and it showed comparable levels to the specification, proving that Krakatoa is capable of mixing, dispensing, and filtering advanced media.
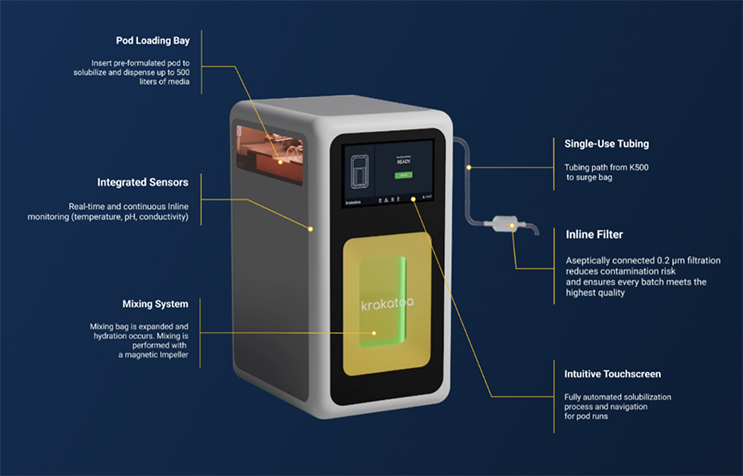
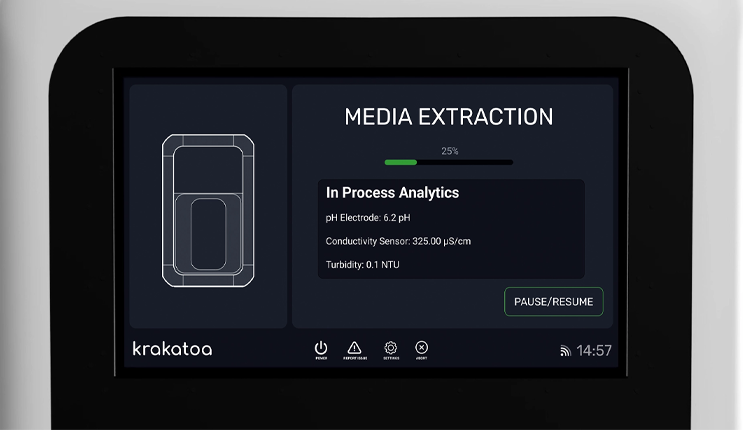
![]() Integrated Analytics and Digital Systems
Integrated Analytics and Digital Systems
Controlled workflows, inline QC sensors for pH, conductivity, and turbidity monitoring, and automatic digital batch record generation ensure the delivery of consistent, reproducible solutions with audit-ready documentation to support GMP-compliant manufacturing for every batch.
Press and Play Automation
Designed to produce sterile GMP cell culture media and buffers on demand, Krakatoa K500 is an easy-to-use, fully closed, single-use manufacturing system that delivers cell culture solutions directly into a surge bag.
![]() STEP 1
STEP 1
Load the Pod and Tubing
Click “Create”. Krakatoa K500 will prompt you to load the preformulated powder pod and connect the single-use tubing.
![]() STEP 2
STEP 2
Automated Solubilization
The Krakatoa K500 reads the QR code on the pod to execute the workflow (hydration, mixing) according to the encoded parameters.
![]() STEP 3
STEP 3
Solution is Dispensed
Liquid media or buffer is extracted through the inline 0.2-micron filter and into a surge bag, where samples can be collected for further testing prior to pumping into the bioreactor.

STEP 1
Insert and Connect Pod
Insert single-use pod into pod loading bay. Pod is equipped with aseptic connectors and QR identification.
STEP 2
Automated Mixing
Inner bag expands and media or buffer is mixed to full volume per encoded parameters.
STEP 3
In-process Analytics
Continuous inline monitoring of pH, conductivity, and turbidity.
STEP 4
Sterile Filtration
Aseptically connected 0.2 μm filtration directly to surge bag.
STEP 5
Sample Verification
Draw analytical samples from surge bag.
STEP 6
Bioreactor
Pump sterile solution directly from surge bag into the bioreactor.
Cell culture media and buffers can account for up to 25–30% of your therapy’s total COGS.
By shifting to powder-based, on-demand manufacturing, the Krakatoa K500 substantially lowers media- and buffer-related costs and shortens lead time, while also helping to make therapies become more accessible to patients.
Built for Modern Biomanufacturing Organizations
The Krakatoa K500 transforms media and buffers from a supply-chain and cost liability into an on-demand manufacturing capability. By decentralizing production and embedding automation, quality, and traceability at the point of use, the Krakatoa K500 enables faster timelines, lower risk, and greater operational efficiency.
Reduce Labor and Variability
Automation replaces manual preparation, mixing, and documentation. With minimal hands-on time and standardized execution, the Krakatoa K500 reduces labor costs, variability, and the risk of costly batch deviations.
Eliminate Supply Chain Bottlenecks
Powder-based pods reduce the number of consumables required and eliminate cold-chain transport and storage. On-demand production removes supplier lead times and reduces incoming QC, storage, and management.
Streamline Quality Compliance
Integrated batch records, inline monitoring (pH, conductivity, turbidity), sterile filtration, and closed, single-use workflows reduce manual documentation and deviations. This helps control the downstream cost of quality compliance, enables traceability and confidence in batch quality, and provides immediate audit-ready documentation.
Reduce Environmental Footprint
By eliminating cold-chain shipping and long-term refrigerated storage, the Krakatoa K500 significantly reduces energy consumption and emissions, supporting both sustainability goals and long-term cost efficiency.
The Power of the Pod
 Media and buffer formulations are prepackaged and delivered ready-to-use in standardized pods designed for flexibility and scale. Two pod sizes support any production volume from 35–100 liters or 175–500 liters. QR-code-driven execution enables seamless operation with full digital batch traceability, simplifying workflows while ensuring consistency and control.
Media and buffer formulations are prepackaged and delivered ready-to-use in standardized pods designed for flexibility and scale. Two pod sizes support any production volume from 35–100 liters or 175–500 liters. QR-code-driven execution enables seamless operation with full digital batch traceability, simplifying workflows while ensuring consistency and control.
Unlock Your Manufacturing Potential
Reshape how biomanufacturing operates.
The Krakatoa K500 is a mobile, self-contained system that brings reproducible media and buffer production directly to the point of use, simplifying operations while increasing control, consistency, and speed.
- Intuitive, user-friendly setup with less than 15 minutes of hands-on time
- Produce up to 500 liters of media or buffers in under 3 hours
- Fully enclosed, single-use design reduces contamination risk and operator exposure
- Automated execution ensures consistent throughput and run-to-run reproducibility
- Real-time process monitoring and automated digital batch records ensures consistency and supports quality compliance
Custom Media and Buffer Development Services
Our formulation services ensure your media and buffers are optimized for powder-based, on-demand manufacturing on the Krakatoa K500. Our scientists work directly with your team to evaluate existing formulations or develop new ones.
- Media and buffer development or optimization tailored to your cell type and process
- Powder-ready formulation design for reliable hydration, mixing, and filtration
- Compatibility testing to ensure consistent execution on the Krakatoa K500
- Seamless tech transfer from development into GMP-compliant formulations

 Integrated Analytics and Digital Systems
Integrated Analytics and Digital Systems STEP 1
STEP 1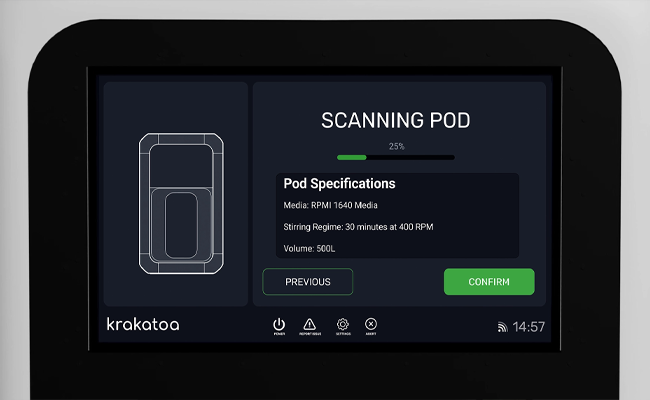 STEP 2
STEP 2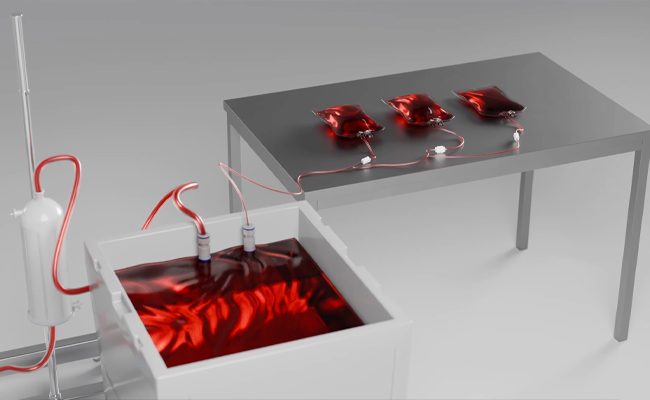 STEP 3
STEP 3