AI, Digitalization, And In Vivo Programming Redefine Cell And Gene Therapy
By Ravi Maharjan, research professor, Yonsei University, and cofounder, Global Reference Laboratories Pvt. Ltd.

The landscape of cell and gene therapy (CGT) is undergoing a significant transformation, driven by the convergence of artificial intelligence (AI), synthetic biology, and digitalization. Initially, the success of CAR-T therapies revolutionized oncology; however, these treatments faced challenges, such as complex manufacturing processes, centralized viral transduction, long lead times, and high costs. In response to these hurdles, the industry is now shifting the discovery and early-stage development phases from complex ex vivo cell engineering toward in vivo programming and molecular design driven by AI, fundamentally reshaping how these therapies are developed, manufactured, and delivered.
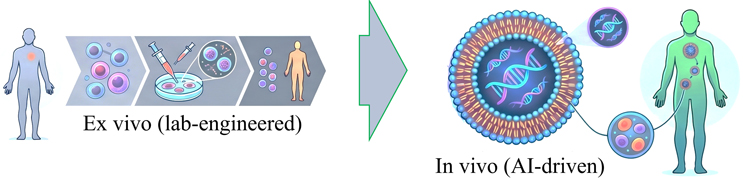
Paradigm Shift Of Cell Products As Genetic Vehicles
The CGT industry is shifting from traditional ex vivo cell engineering to a focus on specialized in vivo gene therapy. In this new paradigm, the therapeutic agent is no longer a living cell engineered in a laboratory but rather a nano-particulate vehicle, lipid nanoparticle (LNP) or adeno-associated virus (AAV), which carries genetic instructions directly into the patient’s system. AI algorithms now enable the rational design of these smart genetic vehicles, optimizing their ability to target specific tissues while minimizing off-target effects. Furthermore, advancements in mRNA delivery systems, such as selective endogenous encapsidation for cellular delivery (SEND), enable the direct in situ programming of immune cells. This innovation eliminates the need for cell isolation and reinfusion, potentially allowing for ready-made solutions that simplify manufacturing. This concept represents a fundamental rethinking of how therapies can be delivered, moving away from the limitations of cell-based products.
AI-Driven Design And Molecular Discovery
Recent advancements in AI have progressed beyond mere data analysis to the de novo design of therapeutic components. Tools like AlphaFold have democratized access to high-resolution protein structures, facilitating the design of synthetic Cas nucleases and novel deaminases for base editing. Foundation models such as ESM-3, trained on millions of sequences, serve as generative tools for creating entirely new proteins. In the realm of delivery, the LUMI model aids in the discovery of novel LNPs for in vivo RNA-based gene editors. Additionally, the AGILE platform employs deep learning to screen thousands of LNPs, successfully identifying vehicles optimized for specific cell types, such as macrophages.
This novel approach allows early development scientists to bypass the biological variability often associated with patient-derived cells, which is a significant issue in earlier clinical treatments. This redirected focus on non-viral gene delivery systems offers several advantages in the field of medicine. LNPs are associated with reduced immunogenicity, as non-viral systems minimize the immune response risk that might complicate the treatment efficacy. By utilizing LNPs, the industry can design and scale products with a lower immunogenic risk compared to traditional viral vectors. Their scalability allows large-scale production with consistent quality. Furthermore, these LNPs can be engineered for targeted delivery, enabling the precise transport of genetic material to specific cell types, thereby enhancing therapeutic outcomes.
Breakthroughs In Genome Editing And Targeted Internalization
The integration of AI and machine learning tools in genome editing technologies such as CRISPR-Cas9 offers unprecedented precision in modifying genetic structures. Next-generation tools like prime and base editing are capable of the single-nucleotide modifications without inducing double-strand breaks (DSBs), thereby reducing the risks of genomic instability. Recent achievements in prime editing have demonstrated efficiencies exceeding 75% in ex vivo modified hematopoietic stem cells. AI-assisted tools, such as DeepSpCas9 and CRISPRon, enhance safety by providing high accuracy in predicting on-target activity and minimizing off-target effects. Moreover, CRISPR-associated transposases (CASTs) enable the site-specific integration of large genetic components with high efficiency, all without creating DSBs.
Additionally, the integration of AI/ML models and specific neural networks into the process has led to remarkable improvements in targeted internalization and encapsulation efficiency, achieving over 90%. Among various AI/ML models, SHapley Additive exPlanations (SHAP) value-driven pKa tuning has emerged as a preferred method. This approach specifically targets the critical pH range of 4.2 to 6.5, optimizing the performance of LNPs for enhanced therapeutic applications. Targeted internalization is a significant advancement in drug discovery, where the monoclonal antibodies are conjugated to the target specific CD3, CD4, or CD8 proteins. This precise docking mechanism allows targeted delivery directly into a specific immune subset within the bloodstream, thereby enhancing therapeutic efficacy of the delivered payload. Alternatively, LNPs can be specifically targeted to the receptors like NKp46, which enable direct delivery of mRNA payload to natural killer (NK) cells.
Smart Biomanufacturing And Digitalization
The adoption of Pharma 4.0 principles is revolutionizing CGT manufacturing, leading to the emergence of smarter biomanufacturing. By integrating devices into digital networks and converting data into searchable formats, manufacturers can achieve real-time monitoring of complex biological processes. Machine learning models for smart perfusion can optimize media usage, achieving reductions of 35% to 50% while simultaneously increasing cell expansion rates. Additionally, digital twins allow for model-based simulations that predict future behaviors and apply optimized conditions in real time.
Synthetic Biology And Logic-Gated Therapeutics
Synthetic biology provides a hierarchical framework capable of sophisticated decision-making. By decomposing biology into modular parts, such as promoters, coding sequences, and protein domains, researchers can construct synthetic circuits. These engineered circuits implement Boolean logic (and, or, not), enabling cells to activate therapeutic responses only when specific combinations of disease signals are present. This approach enhances specificity and reduces toxicity. For instance, synthetic synNotch receptors trigger localized therapeutic programs only upon sensing specific tissue antigens, offering a more holistic recognition strategy for complex diseases like cancer. To further mitigate off-target risks, scientists incorporate microRNA (miRNA) target sites, such as miR-122, into their mRNA payloads. This safety-by-design approach ensures that therapeutic expression is silenced in non-target tissues, even if an LNP reaches the liver, remaining active only within the intended hematopoietic cells. This level of control is crucial for minimizing potential side effects and enhancing patient safety.
Global Access And Point-Of-Care Innovation
Recent advancements are bringing the industry closer to regular access to advanced therapies. Point-of-care (POC) manufacturing, utilizing decentralized models and automated systems like CliniMACS Prodigy, allows for consistent production at hospitals where patients receive personalized treatment. Furthermore, modular facilities, referred to as “CGT in a Box,” consist of mobile, modular cleanrooms that can be deployed globally, reducing the burden on patients and eliminating geographic barriers to treatment. The approval of CRISPR-based therapies for sickle cell disease marks a historic milestone for the field. Moreover, AI accelerates routine tasks and improves data analysis. This allows us to devote more time and resources to addressing complex problems, ultimately leading to faster and more effective therapeutic innovations.
Challenges And The Path Forward
Despite these rapid advancements, significant challenges still remain. The quality and standardization of data are critical; robust AI models require large, high-quality, and normalized data sets. The industry must prioritize a standardized language to ensure that data is searchable across different platforms. Additionally, regulatory evolution is necessary. Agencies like the FDA and EMA have published draft guidelines on the use of AI in CGT life cycles, but a risk-based credibility assessment framework is essential for continuously learning AI systems, which these guidelines currently lack. Ethical considerations surrounding editing, data privacy, and equitable access must also be addressed to maintain public trust.
By merging precision genetic engineering with the scalability of nanomedicine and the predictive power of AI, scientists are reshaping the future of human health. This shift toward smart, targeted genetic vehicles promises to make advanced therapies not only more effective but also safer and more accessible for patients worldwide.
About The Author
 Ravi Maharjan, Ph.D., is a research professor at Yonsei University, Korea, and an associate editor for the IEEE Transactions on NanoBioscience. With experience in pharmaceutical development and nanotechnology, Maharjan specializes in RNA-LNP vaccines, CGT therapy, microfluidics, and biophysical characterization of complex biologics. His expertise spans the integration of AI/ML applications in drug discovery, including small molecule optimization and solubility enhancement through advanced thermodynamic modeling. Prior to his current role at Yonsei, he was a research professor at Dongguk University. Maharjan is a recognized expert in formulation and stability studies, with considerable experience in lyophilization, spray drying, and 3D printing for pharmaceutical applications.
Ravi Maharjan, Ph.D., is a research professor at Yonsei University, Korea, and an associate editor for the IEEE Transactions on NanoBioscience. With experience in pharmaceutical development and nanotechnology, Maharjan specializes in RNA-LNP vaccines, CGT therapy, microfluidics, and biophysical characterization of complex biologics. His expertise spans the integration of AI/ML applications in drug discovery, including small molecule optimization and solubility enhancement through advanced thermodynamic modeling. Prior to his current role at Yonsei, he was a research professor at Dongguk University. Maharjan is a recognized expert in formulation and stability studies, with considerable experience in lyophilization, spray drying, and 3D printing for pharmaceutical applications.
