Activate Process Intensification Strategies To Increase Productivity And Lower Manufacturing Footprint

In the pursuit of more efficient and cost-effective biomanufacturing strategies, drug developers and CDMOs are exploring adaptive production techniques that can modernize legacy facilities without expanding footprint. As complex modalities move toward commercialization, the need for flexible facilities capable of supporting multiple modalities and fluctuating demand continues to grow. Amid pressure to cut drug costs, improve process sustainability, and compete with biosimilars, process intensification (PI) has emerged as a compelling option to increase yields and future-proof operations.
Crucially, PI is not limited to mAbs; it can be implemented across a wide range of modalities, including emerging molecules, like multi-specifics, where stability concerns and degradation risks may necessitate perfusion-based processes to achieve target yields. As sponsors and CDMOs prepare for the next phase of biomanufacturing, many are examining the market forces driving PI adoption, the practical steps to implementation, and the tradeoffs associated with continuous processing.
Why PI, And Why Now?
The approach to mAb manufacturing has steadily evolved since its inception:
- 1990-2000 (COGS ~1,000 €/g for mAbs): Manufacturers relied on inefficient batch-mode processes in large, stainless-steel tanks, resulting in low titers, high energy use, long lead times, and limited flexibility
- 2000-2020 (COGS < 200 €/g for mAbs): This era marked a critical shift toward small batch processes using single-use technology, delivering higher titers, lower capital investment, and greater flexibility
- 2020 and beyond (COGS < 50 €/g for mAbs): This period reflects a growing pivot toward intensified and continuous processes enabled by automation and advanced technologies, leading to higher productivity, improved sustainability, and reduced CO2 emissions.
This evolution highlights that advances in perfusion bioreactors, membrane chromatography, data analytics, and automation have made PI a more viable operational model. At its core, PI aims to reduce facility footprint and operational costs while maintaining or improving product quality. Many CDMOs are adopting intensified processes to increase throughput and ROI per square foot. With greater productivity and better equipment, CDMOs can offer clients competitive pricing, rapid changeovers, and multi-modality manufacturing within the same infrastructure.
By enabling more efficient use of time, facility space, and equipment, PI allows organizations to do more with less. Single-use technology further enhances flexibility by enabling rapid adjustments to accommodate changing production needs. As a result, PI is increasingly viewed as a pathway to lower COGS and more resilient manufacturing operations.
What Are Industry Stakeholders Saying About PI?
PI is already being used to manufacture 25+ commercially approved drugs across 80+ manufacturing facilities. Equipment demand reflects this trend; at Sartorius, 1 in 3 of our bioreactors are sold with perfusion capability and more than 100 customers currently use our intensified chromatography solutions.
Industry leaders also point to PI’s broader strategic value. According to Kevin Brower, Global Head of Purification Development at Sanofi, “PI is critically important for Sanofi and the broader industry to decrease costs, increase access to medicine for patients, and meet sustainability goals.” Meanwhile, Himanshu Gadgil, Director and CSO of Enzene Biosciences, reports a tenfold increase in productivity, a 50% reduction in COGS, and a significantly lower carbon footprint using a fully connected continuous process.
How Is PI Implemented?
For most organizations, PI is not an all or nothing pursuit. Instead, it is introduced in stages. Figure 1 outlines the four levels to PI adoption:
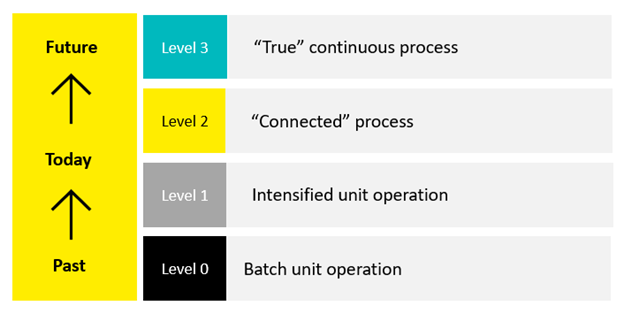
Figure 1. The pathway to PI activation.
Level 0 represents the industry’s traditional approach: upstream and downstream processing in fed-batch mode. Level 3 represents a truly continuous process where unit operations flow seamlessly into one another. However, even partial adoption, such as Level 1 or Level 2 semi-continuous processes, can produce measurable reductions in footprint, costs, emissions, and productivity.
As industry stakeholders evaluate PI, many are taking a holistic view of upstream to downstream operations to identify potential bottlenecks and mitigate risks. Continuous processes are typically implemented at development scale first, allowing teams to generate the data needed to support clinical to commercial manufacturing. CDMOs are also prioritizing equipment that supports automation, flexibility, and interchangeability, easing implementation, reducing long-term costs, and improving adaptability to different modalities.
To enable digital tech transfer efficiency, facilities should leverage consistent technologies that support seamless transitions. As PI best practices for quality control and regulatory compliance continue to mature, stakeholders can expect to see faster implementation and deployment in the future. Industry working groups and collaborative initiatives will play a key role by sharing ideas, developing guidance, and providing training resources to help teams prepare for PI implementation.
What Are The Barriers To Implementing PI?
Despite its advantages, PI represents a significant shift from traditional manufacturing models. Implementation can appear risky or difficult in a conservative, highly regulated industry, particularly when major capital investment is required. Retrofitting an existing facility to support continuous processes is complex, and building a new automated plant is costly. Often, legacy process control systems are not compatible with continuous operations, requiring upgrades before PI can be introduced.
Beyond the necessary resources, there is also the time investment needed to successfully activate and scale a continuous process. If leadership is evaluating a program with an aggressive two-year timeline to market, the obvious route may still be a well-understood fed-batch process. As a result, many CDMOs are still determining how best to balance PI adoption with the need for scalability, speed to market, and regulatory confidence.
What Are The Best Use Cases For PI?
This question can only be answered by those familiar with a company’s modalities, needs, and cost constraints. Sponsors and CDMOs must collaborate to identify feasible entry points that deliver clear value while building experience with intensified processes. In addition to PI’s ability to support unstable multi-specific products, it can also be advantageous in biosimilar development, where establishing early proof-of-concept helps developers determine whether a product will be viable.
PI can also maximize throughput, enable targeted debottlenecking, and efficiently manage the increasingly high titers coming out of upstream processes. In one customer case study, a facility had limited buffer preparation capacity and facility size constraints. The team leveraged a downstream skid to conduct inline buffer dilution and multi-column chromatography, moving from a batch cadence of every two weeks to every four days.
In another example, a client was working with an unstable, bispecific molecule where affinity capture was not an option and clipping occurred during feedback. By applying PI to the perfusion and chromatography steps, the product was captured as soon as it was produced, preventing clipping and achieving nearly nine times the amount of product from the same bioreactor.
Is PI A Viable Option?
Continuous processing is the next frontier of biomanufacturing. Success, however, requires sustained investment alongside a willingness to adapt to automated, data-driven processes. While the transition can be complex and challenging, due diligence and the right technology can clear a path to PI adoption, reducing COGS and increasing productivity while helping sponsors and CDMOs achieve sustainability targets. Therefore, when evaluating long-term manufacturing strategies, the question is increasingly not whether PI will play a role, but where and when it can deliver the greatest operational and economic impact.
About Sartorius
At Sartorius, we empower scientists and engineers to simplify and accelerate progress in life science and bioprocessing, enabling the development of new and better therapies and more affordable medicine.
